Friday, December 31, 2010
A Frigorific New Year!
The temperature of course can't get much above freezing with all this ice all over everything. Looks like a frigorific new year.
Thursday, December 23, 2010
Passed round one
I've been invited to give a short talk on my research to the committee deciding which applicants will be invited to form Research Groups. The interview will be mid-February in Berlin, and I should know very shortly after that if my application was successful.
This means I have a month and a half to write a talk, make impressive figures and graphs, and so forth. The hardest part for me in writing the talk is figuring out who my audience is. The committee that wants to interview me is from the Human Sciences Section. This includes institutes focusing on anthropology, economics, art-history, law, religious and ethnic studies, linguistics, sociology, the history of science and the study of cognition. It also includes demography, evolutionary anthropology and a fair bit of other natural sciences. I can't simply give the same talk to a group of biologists and demographers that I would give to a committee of lawyers and ethnographers. I don't yet know if the committee as a whole will be judging my talk, in which case I have to assume a great diversity of background, or if they will have a smaller group of specialists assigned to each talk. I can give a good talk either way, but not both simultaneously. I've got some thinking to do.
Friday, December 17, 2010
Christmas Party of Science
My favorite part of the party was the Powerpoint Karaoke. This is a game which could be popular only with academics. Each player gets up to give a talk with power-point slides, which sounds boring, until you consider that he has never seen these slides before, and they are on a topic he likely knows nothing about. There were three contestants, including Santa, and each of us prepared a set of slides another one of us had to speak on. We were not kind to each other. My friend Jon spoke first, on a set of slides I had made, on sesquipedalianisms. I had the longest and most complicated words in several languages under the bright red heading “SAY THESE WITH ME!!.” He made a valiant attempt. Next I got up, in costume, and my slides were entirely in Greek. I recognized the alphabet, but had no idea what any of the words said, and there were no pictures. Lucky for me, most of my audience also didn’t read Greek, so I pretended that I knew exactly what the slides said, and that they were designed to correct common misconceptions about Santa (e.g., Santa does not in fact employ any reindeer, as their odor offends his sensitive nose.) The talk went quite well, and only afterwards did I find out that it was a presentation in Greek on numerical modeling. Finally, my friend Mikko got up and gave a talk on obscure economic phenomena he knew nothing about. He did a creditable job of pretending he knew what he was saying, or rather of being so precise in his vagaries that it was a believable if entirely uninformative talk. It was the first time I’d played this game, but I think it will not be the last.
Frohe Weinachten! Ho Ho ho!!!
Saturday, November 27, 2010
Could be good
If he ends up writing about my work it will bring my mother great bragging points with her cousins whether he gets it right or not, but I would be greatly annoyed if he slaughters it.
Competing by not competing
Despite this, I am setting out to write a human demography paper, with little if any evolution in it. I can do this with some confidence because, as far as I know, I am working in an area that has been almost completely overlooked, and therefore I have no competition. Most anyone with a solid demography background could do a better job of what I want to do than I can, and I really couldn't compete. But because they haven't bothered, I don't have to compete. I can do a decent job and hopefully publish in a good journal without worrying about the competition, because I believe there to be none.
In my recent grant application, I wrote, "My work falls between demography and evolution, outside the well explored territory of either. Work within evolutionary demography tends to focus on senescence and reproduction; I have intentionally eschewed these to seek the question others have avoided. This is a high risk strategy; my work does not fit neatly into any one topic-specific journal or discipline. However this unconventional approach has the opportunity to found a new direction of study and investigate the most important questions therein." This is a polite way of saying that I intentionally avoid competition by seeking out the questions others have ignored, or deemed less interesting. Most successful scientists are successful because they look for opportunities to do what others aren't doing. I don't know to what extent other seek out whole areas that others haven't bothered with. I also don't know how successful or sustainable a strategy this is likely to be in the long run. After all, the success of the work will ultimately be measured by how many other people get interested in it, and try to improve upon it. Successful work, by definition, must therefore attract competition. So if I want to be successful, but continue not having competitors, I’ll need to move on to some other under-appreciated topic fairly quickly. As I rather like the topic I’m currently seeding, I may just have to put up with some competition, and try to stay ahead of them. having my own research group would be a great help in this. Not being an expert demographer is less of a problem if you have an expert demographer on staff.
Sunday, November 21, 2010
More Tardigrades
Anyway, here, by popular demand, are a couple more pictures.
Note the long curved claws at the ends of the toes, much like a bear has.
Saturday, November 20, 2010
Fussy Hydra babies
I find myself looking forward to having rotifers in the lab again. They are just so familiar at this point.
Friday, November 19, 2010
The Royal Society has informed me
Focus
Science, like most complex tasks, is much easier to make progress on if you know exactly what you are doing, and you have everything you need to do it ready. Over the last couple of months, I got a lot done because I focused on finishing my grant application, and on getting out the two papers I needed to finish to go with my application. Now the application is in, the review paper is accepted, and the methods paper is submitted, and I must find a new focus to guide me. Over the last couple of days I have been scattering my time and thoughts between a dozen projects, and not really making any progress on any of them. I am giving myself until Monday to decide what the new plan is.
Thursday, November 18, 2010
Tardigrades!
What is so cool about tardigrades? They can survive total dehydration, exposure to vacuum, radiation, freezing, extreme heat, conservative radio personalities, you name it. They can persist for a decade dried out with no signs of life, then just add water and they reanimate themselves. They are rolly and round and shaped kind of like six-legged teddy bears with two more legs growing out of their tails. This appearance has earned them the nickname ‘water bears.’
This afternoon I wanted to see if the hydra I am keeping in the lab would eat rotifers, because the Artemia I am giving them now are a bit too big for them, and rotifers a tiny. I found some in a puddle outside the institute, and in the same puddle, I found tardigrades. I had never knowingly seen a live tardigrade before, so I took a good long look at them under the microscope. These ones were well under a millimeter long, but I tried to get pictures.
They are small enough that really good photos would require a different microscope, but here are two.
This is a tardigrade from above. You can't really see how cute they are from above.
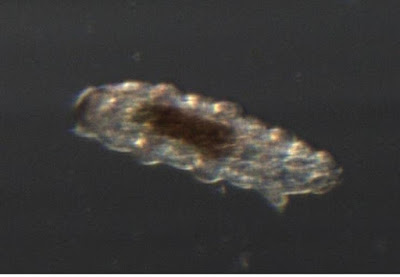
This is the shed skin of a tardigrade.
Just as snakes have to shed their stiff skins occasionally, many animals with exoskeletons molt that shell occasionally. All the arthropods (insects, spiders, crustaceans, crabs etc) will shed their skins, and so will tardigrades. Tardigrades are not arthropods, but are their closest realtive except for the velvet worms. This skin is head-end down. You can clearly see the six legs on the left side, and the two-legged tail at the top-left. I will try to get better pictures of a live critter tomorrow.
Sunday, November 14, 2010
Jon Asks: 2
In the Laura Ingalls Wilder book The Long Winter, Laura's father says that you can predict the severity of a winter by observing the thickness of muskrat nests in the summer. Muskrats, he says, will build thicker nests during the summer if the following winter is going to be relatively colder, and vice versa with thinner nests and relatively warmer winters. Has this folk wisdom been investigated? Is it true? And if so, where can I get my own muskrat colony?
I can't find anything on Google Scholar or Web of Science indicating that anyone has published anything about muskrats and weather prediction, other than this:
Man's natural craving for advance knowledge of coming weather extends thousands of years back of any attempts at scientific weather forecasting. Realizing that he has not the necessary foresight himself, he has imagined animals to be endowed with some peculiar sense which enables them to know, weeks or months ahead, what the weather will be. Thus a large group of animal weather proverbs has come into existence. Millions of people believe that the thickness of fur on a muskrat, or the number of nuts stored by a squirrel, or a supposedly early migration of certain birds, indicates a severe winter. Yet it is certain that animals have no such foresight.
from: Robert DeC. Ward. 1926. The Present Status of Long-Range Weather Forecasting
Proceedings of the American Philosophical Society,Vol. 65, No. 1, pp. 1-14
He provides no evidence to show that they don't, he is just certain. Note that the version you mention has to do with the thickness of the wall of the house, while the version Ward mentions has to do with the thicknesss of their fur. The fur hypothesis would be easier to test, if you were a muskrat hunter. I am frankly doubtful whether he or anyone else has done the work that would be needed to convincingly either story. You would have to measure the wall thickness of bunches of muskrat houses (or the pelt of many muskrats) in the summer. You would have to do this every year for quite a few years in order to make a convincing analysis of the relationship between wall thickness and hardness of winter. You would probably also want to measure various features of the microclimate, the muskrats behavior and physiology, and the local ecology, in order to get some sense of what the mechanism was. You probably would want to measure the pelts and the houses, just to make sure you were measuring the right thing. This is one limitation to testing folk-wisdom. There are often several versions, and it is hard to know if you are testing the right one unless you test all of them, and then you increase your chances of finding a strong correlation just by chance. My best guess is that there is some, but not a lot of, truth to either version of the story. Certainly they could pick up on whatever cues are available that the winter is going to be hard. But like most weather prediction, they probably aren't very accurate, at least not months in advance.
Jon Asks: 1
I've read that fungi are the only organisms that can degrade the longer-chain fibers in wood, such as lignin, and that without saprobic fungi the world would be blanketed in dead, undecayed trees. I see on Wikipedia that it is not literally true that no bacteria can degrade lignins, however, by Wiki's account, it does seem that no known bacteria are very good at it. (http://en.wikipedia.org/wiki/Ligninase) So why would that be the evolutionary case? Bacteria have evolved to break down pretty much everything else on the planet (roughly speaking), and wood has been around for something like 350+ million years. Why would they be such second-rate degraders when it comes to lignin?
This is an interesting question, but not one I can give a very satisfying answer to. Explanations of why something didn't evolve are always fairly speculative. Why no six legged tigers? Why no live-birthing birds? Why no Ents?
So why no lignin devouring bacteria? If they can do it poorly, why not well? Maybe it isn't worth their while to invest in that capacity, as they are always outcompeted by the fungi who can already do it? Maybe they can rely on the fungi to make the enzymes, and then they can just mooch. Perhaps the process of making the necessary enzymes requires separate cellular compartments, which bacteria lack. Maybe the necessary mutations just never occurred, and so couldn't be selected for. Certainly I don't know.
Friday, November 12, 2010
Winter
I have found that the best strategy for keeping my bad hip from minding the weather is to make sure I am absurdly over-dressed. This morning I walked to work, in temperatures above freezing, wearing high insulated shoes, long-underwear, lined pants, a heavy over-shirt, a down parka and thick gloves, hat and scarf. I was sweating the whole time, but my hip felt fine.
All of this does not endear the city to my heart, and yet I hope I will be here another five years. The grant application that is taking most of my time is to establish a research group of my own, funded by the Max Planck Society for five years. There are three responses they could make to my application. They could say no, they could say yes and let me form the research group here, or they could say yes but tell me to form it at a different Max Planck Institute, in a different city (i.e., Cologne).
I indicate in my application that my first choice is to stay here (at MPIDR), not out of love for the climate, but because of MPIDR’s, “strength in evolutionary demography would be a great benefit to me. I have ongoing collaborations with several MPIDR researchers, including members of the Research Groups on Lifecourse Dynamics and Demographic Change, and Modeling the Evolution of Aging, and of the Laboratories of Statistical Demography, Survival and Longevity, and Evolutionary Biodemography. As my project draws on evolutionary and demographic theory, on MPIDR's Human Mortality Database and Biodemographic Database, on laboratory experiments and survey data, MPIDR’s mixture of social science and biology is the ideal environment for me.”
Add to that that we have good friends, Iris enjoys teaching at the University here, we like our colleagues, we have a great apartment, and moving (which we have done far to much of) is a pain in the rear, and I am very much hoping to stay here. That said, if they offer me a group in Cologne, we will certainly go, in the spring.
Thursday, November 11, 2010
Good news
The point of a review article is to give an overview of the field. What is know, what is hypothesized, what are the important questions, where is the field going. I have been thinking about the topic of this one on and off for perhaps seven years, but if you put together all the time I specifically spent on it, it would be about six months of work, most in the last year. Perhaps half of that was spent just searching out the relevant literature. I must have read several thousand article titles, perhaps 500 abstracts, and maybe 200 full papers and book chapters. 91 sources made it into the final paper, and 18 more into the appendixes. The reason I had to do so much preliminary literature searching is that no one has ever written a review on this topic before, and perhaps four of the authors whose ideas and data I draw on had this general topic in mind.
Now, part of publishing with them and most other journals these days is that the paper is embargoed until they say it ain't. Embargoed means I can't tell the press what I found out, or even what the article is about, before the journal has a chance to publish it. But for the curious and bored, I can share the list of articles I reference. Just looking through them gives a sense of the range of journals I was searching in. Archiv fur Hydrobiologie, The American Journal of Tropical Medicine and Hygiene, Entomol. Exp. Appl, American Statistician, Obstetrical & Gynecological Survey, Can. J. Fish. Aquat. Sci., Experimental Gerontology, The Auk, Annals: New York Academy of Sciences, Biol. Reprod, Maturitas, Administrative Science Quarterly, J. Herpetol., Ophelia, Genetica, Journal of the Institute of Actuaries and so on. I am lucky in that I didn't have to actually scan the tables of contents of the several thousand journals that could potentially have had relevant papers. I used Google Scholar, Web of Science and other literature searching tools. I have no idea how they did this sort of thing before the internet.
Like a good playbill, in order of appearance:
Main Article:
1. Medawar, P. B. 1952 An unsolved problem of biology. London: HK Lewis.
2. Metcalf, C. J. E. & Pavard, S. 2007 Why evolutionary biologists should be demographers. Trends Ecol. Evol. 22, 205-212.
3. Wachter, K. W. 2008 Biodemography comes of age. Demographic Research 19, 1501-1512.
4. Vaupel, J. W. 2010 Biodemography of human ageing. Nature 464, 536-542.
5. Moorad, J. A. & Promislow, D. E. L. 2009 What can genetic variation tell us about the evolution of senescence? Proc. R. Soc. Lond., Ser. B: Biol. Sci.
6. Péron, G., Gimenez, O., Charmantier, A., Gaillard, J. M. & Crochet, P. A. 2010 Age at the onset of senescence in birds and mammals is predicted by early-life performance. Proceedings of the Royal Society B.
7. Young, H. 1963 Age-specific mortality in the eggs and nestlings of blackbirds. The Auk 80, 145-155.
8. Deevey, E. S. 1947 Life tables for natural populations of animals. The Quarterly Review of Biology 22, 283-314.
9. Jones, O. R., Gaillard, J. M., Tuljapurkar, S., Alho, J. S., Armitage, K. B., Becker, P. H., Bize, P., Brommer, J., Charmantier, A. & Charpentier, M. 2008 Senescence rates are determined by ranking on the fast–slow life-history continuum. Ecol. Lett. 11, 664-673.
10. Pike, D. A., Pizzatto, L., Pike, B. A. & Shine, R. 2008 Estimating survival rates of uncatchable animals: The myth of high juvenile mortality in reptiles. Ecology 89, 607-611.
11. Carey, J. R., Liedo, P. & Vaupel, J. W. 1995 Mortality dynamics of density in the Mediterranean fruit fly. Experimental Gerontology 30, 605-629.
12. Pletcher, S. D., Macdonald, S. J., Marguerie, R., Certa, U., Stearns, S. C., Goldstein, D. B. & Partridge, L. 2002 Genome-wide transcript profiles in aging and calorically restricted Drosophila melanogaster. Curr. Biol. 12, 712-723.
13. Rose, M. R. 1984 Laboratory evolution of postponed senescence in Drosophila melanogaster. Evolution 38, 1004-1010.
14. Tatar, M., Carey, J. R. & Vaupel, J. W. 1993 Long-term cost of reproduction with and without accelerated senescence in Callosobruchus maculatus: analysis of age-specific mortality. Evolution 47, 1302-1312.
15. Bauer, G. 1983 Age structure, age specific mortality rates and population trend of the freshwater pearl mussel(Margaritifera margaritifera) in North Bavaria. Archiv fur Hydrobiologie 98, 523-532.
16. Styer, L. M., Carey, J. R., Wang, J. L. & Scott, T. W. 2007 Mosquitoes do senesce: departure from the paradigm of constant mortality. The American Journal of Tropical Medicine and Hygiene 76, 111.
17. Sarup, P. & Loeschcke, V. 2010 Life extension and the position of the hormetic zone depends on sex and genetic background in Drosophila melanogaster. Biogerontology, 1-9.
18. Caughley, G. 1966 Mortality patterns in mammals. Ecology 47, 906-918.
19. Barlow, J. & Boveng, P. 1991 Modeling age-specific mortality for marine mammal populations. Mar. Mamm. Sci. 7, 50-65.
20. Spinage, C. A. 1972 African ungulate life tables. Ecology 53, 645-652.
21. Bruderl, J. & Schussler, R. 1990 Organizational mortality: The liabilities of newness and adolescence. Administrative Science Quarterly 35, 530-547.
22. Yang, G. 2007 Life cycle reliability engineering. Hoboken, NJ: John Wiley & Sons Inc.
23. Holliday, R. 2006 Aging is no longer an unsolved problem in biology. Annals: New York Academy of Sciences 1067, 1-9.
24. Hamilton, W. D. 1966 The moulding of senescence by natural selection. J. Theor. Biol. 12, 12-45.
25. Williams, G. C. 1957 Pleiotropy, Natural Selection, and the Evolution of Senescence. Evolution 11, 398-411.
26. Baudisch, A. 2008 Inevitable aging?: contributions to evolutionary-demographic theory: Springer Verlag.
27. Kirkwood, T. B. L. 1990 The disposable soma theory of aging. In Genetic effects on aging (ed. D. E. Harrison), pp. 9–19. Caldwell, NJ: Telford Press.
28. Holman, D. J. & Wood, J. W. 2001 Pregnancy loss and fecundability in women. In Reproductive ecology and human evolution (ed. P. T. Ellison), pp. 15–38. Hawthorne, NY: Aldine de Gruyter.
29. Woods, R. 2009 Death before Birth. Oxford: Oxford University Press.
30. O'Connor, K. A., Holman, D. J. & Wood, J. W. 1998 Declining fecundity and ovarian ageing in natural fertility populations. Maturitas 30, 127-136.
31. Kruger, D. J. & Nesse, R. M. 2004 Sexual selection and the Male:Female Mortality Ratio. Evolutionary Psychology 2, 66-85.
32. Bonduriansky, R. & Brassil, C. E. 2002 Senescence: rapid and costly ageing in wild male flies. Nature 420, 377.
33. McDonald, D. B., Fitzpatrick, J. W. & Woolfenden, G. E. 1996 Actuarial senescence and demographic heterogeneity in the Florida Scrub Jay. Ecology 77, 2373-2381.
34. Roach, D. A., Ridley, C. E. & Dudycha, J. L. 2009 Longitudinal analysis of Plantago: age-by-environment interactions reveal aging. Ecology 90, 1427.
35. Nussey, D. H., Coulson, T., Festa-Bianchet, M. & Gaillard, J. M. 2008 Measuring senescence in wild animal populations: towards a longitudinal approach. Funct. Ecol. 22, 393-406.
36. Sheader, M. 2009 The reproductive biology and ecology of Gammarus duebeni (Crustacea: Amphipoda) in southern England. Journal of the Marine Biological Association of the UK 63, 517-540.
37. Milnes, M. R., Bryan, T. A., Katsu, Y., Kohno, S., Moore, B. C., Iguchi, T. & Guillette, L. J. 2008 Increased posthatching mortality and loss of sexually dimorphic gene expression in alligators (Alligator mississippiensis) from a contaminated environment. Biol. Reprod. 78, 932.
38. Anderson, D. J. 1990 Evolution of obligate siblicide in boobies. 1. A test of the insurance-egg hypothesis. Am. Nat. 135, 334-350.
39. Strandberg, R., Klaassen, R. H. G., Hake, M. & Alerstam, T. 2009 How hazardous is the Sahara Desert crossing for migratory birds? Indications from satellite tracking of raptors. Biol. Lett.
40. Moss, C. J. 2006 The demography of an African elephant (Loxodonta africana) population in Amboseli, Kenya. J. Zool. 255, 145-156.
41. Sumich, J. L. & Harvey, J. T. 1986 Juvenile mortality in gray whales (Eschrichtius robustus). J. Mammal. 67, 179-182.
42. Anderson, J. T. 1988 A review of size dependent survival during pre-recruit stages of fishes in relation to recruitment. Journal of Northwest Atlantic Fisheries Science 8, 55-66.
43. Gislason, H., Daan, N., Rice, J. C. & Pope, J. G. 2010 Size, growth, temperature and the natural mortality of marine fish. Fish Fish. 11, 149-158.
44. Baron, J. P., Le Galliard, J. F., Tully, T. & Ferrière, R. 2010 Cohort variation in offspring growth and survival: prenatal and postnatal factors in a late-maturing viviparous snake. J. Anim. Ecol. 79, 640-649.
45. Kushlan, J. A. & Jacobsen, T. 1990 Environmental variability and the reproductive success of Everglades alligators. J. Herpetol. 24, 176-184.
46. Congdon, J. D., Dunham, A. E. & Van Loben, R. C. 1993 Delayed sexual maturity and demographics of Blanding's turtles (Emydoidea blandingii): implications for conservation and management of long-lived organisms. Conserv. Biol. 7, 826-833.
47. Petranka, J. W. 1985 Does age-specific mortality decrease with age in amphibian larvae? Copeia 1985, 1080-1083.
48. Wilbur, H. M. 1980 Complex life cycles. Annu. Rev. Ecol. Syst. 11, 67-93.
49. Miller, T. J., Crowder, L. B., Rice, J. A. & Marschall, E. A. 1988 Larval size and recruitment mechanisms in fishes: toward a conceptual framework. Can. J. Fish. Aquat. Sci. 45, 1657-1670.
50. Rabinovich, J. E., Nieves, E. L. & Chaves, L. F. 2010 Age-specific mortality analysis of the dry forest kissing bug, Rhodnius neglectus. Entomol. Exp. Appl. 135, 252-262.
51. Chapman, A. R. O. 1986 Age versus stage: An analysis of age-and size-specific mortality and reproduction in a population of Laminaria longicruris Pyl. J. Exp. Mar. Biol. Ecol. 97, 113-122.
52. Cole, K. M. & Sheath, R. G. 1990 Biology of the red algae: Cambridge Univ Pr.
53. Hett, J. M. 1971 A dynamic analysis of age in sugar maple seedlings. Ecology 52, 1071-1074.
54. Leak, W. B. 1975 Age distribution in virgin red spruce and northern hardwoods. Ecology 56, 1451-1454.
55. Harcombe, P. A. 1987 Tree life tables. Bioscience 37, 557-568.
56. Leverich, W. J. & Levin, D. A. 1979 Age-specific survivorship and reproduction in Phlox drummondii. Am. Nat. 113, 881-903.
57. Hanley, M. E., Fenner, M. & Edwards, P. J. 1995 The effect of seedling age on the likelihood of herbivory by the slug Deroceras reticulatum. Funct. Ecol. 9, 754-759.
58. Gosselin, L. A. & Qian, P. Y. 1997 Juvenile mortality in benthic marine invertebrates. Mar. Ecol. Prog. Ser. 146, 265-282.
59. Hallock, P. 1985 Why are larger foraminifera large? Paleobiology 11, 195-208.
60. Levin, L., Caswell, H., Bridges, T., DiBacco, C., Cabrera, D. & Plaia, G. 1996 Demographic responses of estuarine polychaetes to pollutants: life table response experiments. Ecol. Appl. 6, 1295-1313.
61. Greeff, J. M., Storhas, M. G. & Michiels, N. K. 1999 Reducing losses to offspring mortality by redistributing resources. Funct. Ecol., 786-792.
62. Klug, H. & Bonsall, M. B. 2007 When to care for, abandon, or eat your offspring: the evolution of parental care and filial cannibalism. The American Naturalist 170, 886.
63. Manica, A. 2002 Filial cannibalism in teleost fish. Biological Reviews 77, 261-277.
64. Lee, R. D. 2003 Rethinking the evolutionary theory of aging: Transfers, not births, shape senescence in social species. Proc. Natl. Acad. Sci. USA 100, 9637-9642.
65. Rogers, A. R. 2003 Economics and the evolution of life histories. Proc. Natl. Acad. Sci. USA 100, 9114-9115.
66. Lee, R. 2008 Sociality, selection, and survival: Simulated evolution of mortality with intergenerational transfers and food sharing. Proc. Natl. Acad. Sci. USA 105, 7124-7128.
67. Rumrill, S. S. 1990 Natural mortality of marine invertebrate larvae. Ophelia 32.
68. Jones, D. & Göth, A. 2008 Mound-builders. Collingwood VIC: CSIRO.
69. Chu, C. Y. C., Chien, H. K. & Lee, R. D. 2007 Explaining the optimality of U-shaped age-specific mortality. Theor. Popul. Biol. 73, 171-180.
70. Biro, P. A., Abrahams, M. V., Post, J. R. & Parkinson, E. A. 2006 Behavioural trade-offs between growth and mortality explain evolution of submaximal growth rates. Ecology 75, 1165-1171.
71. Sterck, F. J., Poorter, L. & Schieving, F. 2006 Leaf traits determine the growth-survival trade-off across rain forest tree species. Am. Nat. 167, 758-765.
72. Soler, J. J., de Neve, L., Pérez-Contreras, T., Soler, M. & Sorci, G. 2003 Trade-off between immunocompetence and growth in magpies: an experimental study. Proc. R. Soc. Lond., Ser. B: Biol. Sci. 270, 241-248.
73. Mangel, M. & Stamps, J. 2001 Trade-offs between growth and mortality and the maintenance of individual variation in growth. Evol. Ecol. Res. 3, 583-593.
74. Johnson, D. W. & Hixon, M. A. 2010 Ontogenetic and spatial variation in size-selective mortality of a marine fish. J. Evol. Biol. 23, 724-737.
75. Munch, S. B. & Mangel, M. 2006 Evaluation of mortality trajectories in evolutionary biodemography. Proc. Natl. Acad. Sci. USA 103, 16604-16607.
76. von Bertalanffy, L. 1957 Quantitative laws in metabolism and growth. The Quarterly Review of Biology 32, 217-231.
77. Vaupel, J. W., Manton, K. G. & Stallard, E. 1979 The impact of heterogeneity in individual frailty on the dynamics of mortality. Demography 16, 439-454.
78. Vaupel, J. W. & Yashin, A. I. 1985 Heterogeneity's ruses: some surprising effects of selection on population dynamics. American Statistician 39, 176-185.
79. Charlesworth, B. 2001 Patterns of age-specific means and genetic variances of mortality rates predicted by the mutation-accumulation theory of ageing. J. Theor. Biol. 210, 47-65.
80. Mueller, L. D. & Rose, M. R. 1996 Evolutionary theory predicts late-life mortality plateaus. Proc. Natl. Acad. Sci. USA 93, 15249-15253.
81. Moorad, J. A. & Promislow, D. E. L. 2008 A theory of age-dependent mutation and senescence. Genetics 179, 2061-2073.
82. Gong, Y., Thompson Jr, J. N. & Woodruff, R. C. 2006 Effect of deleterious mutations on life span in Drosophila melanogaster. Journals of Gerontology Series A: Biological and Medical Sciences 61, 1246-1252.
83. Pletcher, S. D., Houle, D. & Curtsinger, J. W. 1998 Age-specific properties of spontaneous mutations affecting mortality in Drosophila melanogaster. Genetics 148, 287-303.
84. Yampolsky, L. Y., Pearse, L. E. & Promislow, D. E. L. 2000 Age-specific effects of novel mutations in Drosophila melanogaster I. Mortality. Genetica 110, 11-29.
85. Wexler, N. S. 2004 Venezuelan kindreds reveal that genetic and environmental factors modulate Huntington's disease age of onset. Proc. Natl. Acad. Sci. USA 101, 3498-3503.
86. Wright, S. 1929 Fisher's theory of dominance. Am. Nat. 63, 274-279.
87. Orr, H. A. 1991 A test of Fisher's theory of dominance. Proc. Natl. Acad. Sci. USA 88, 11413-11415.
88. Arbeitman, M. N., Furlong, E. E. M., Imam, F., Johnson, E., Null, B. H., Baker, B. S., Krasnow, M. A., Scott, M. P., Davis, R. W. & White, K. P. 2002 Gene expression during the life cycle of Drosophila melanogaster. Science 297, 2270.
89. Martinez, D. E. 1998 Mortality patterns suggest lack of senescence in hydra. Experimental Gerontology 33, 217-225.
90. Bakketeig, L. S., Seigel, D. G. & Sternthal, P. M. 1978 A fetal-infant life table based on single births in Norway, 1967-1973. Am. J. Epidemiol. 107, 216.
91. Human Life-Table Database. 2010 www.lifetable.de: Max Planck Institute for Demographic Research, Institut national d'études démographiques and UC Berkeley
Appendix 1.
1. Bourgeois-Pichat, J. 1946 De la mesure de la mortalité infantile. Population 1, 53-68.
2. Bourgeois-Pichat, J. 1951 La mesure de la mortalité infantile. II. Les causes de décès. Population 6, 459-480.
3. Wilmoth, J. R. 1997 In search of limits. In Between Zeus and the salmon: The biodemography of longevity (ed. K. W. Wachter), pp. 38-64. Washington, DC: National Academies Press.
4. Carnes, B. A., Holden, L. R., Olshansky, S. J., Witten, M. T. & Siegel, J. S. 2006 Mortality partitions and their relevance to research on senescence. Biogerontology 7, 183-198.
5. Siler, W. 1979 A competing-risk model for animal mortality. Ecology 60, 750-757.
6. Gage, T. B. 1998 The comparative demography of primates: with some comments on the evolution of life histories. Annual Review of Anthropology 27, 197-221.
7. Heligman, L. & Pollard, J. H. 1980 The age pattern of mortality. Journal of the Institute of Actuaries 107, 49–80.
8. Lee, E. T. & Go, O. T. 1997 Survival analysis in public health research. Annu. Rev. Public Health 18, 105-134.
9. Ndirangu, J., Newell, M. L., Tanser, F., Herbst, A. J. & Bland, R. 2010 Decline in early life mortality in a high HIV prevalence rural area of South Africa: evidence of HIV prevention or treatment impact? AIDS 24, 593.
Appendix 2
1. Bishop, M. W. H. 1964 Paternal contribution to embryonic death. Reproduction 7, 383-396.
2. Wilcox, A. J., Weinberg, C. R., O'Connor, J. F., Baird, D. D., Schlatterer, J. P., Canfield, R. E., Armstrong, E. G. & Nisula, B. C. 1989 Incidence of early loss of pregnancy. Obstetrical & Gynecological Survey 44, 147.
3. Boklage, C. E. 1990 Survival probability of human conceptions from fertilization to term. International Journal of Fertility 35, 75-94.
4. O'Connor, K. A., Holman, D. J. & Wood, J. W. 1998 Declining fecundity and ovarian ageing in natural fertility populations. Maturitas 30, 127-136.
5. Hassold, T. & Hunt, P. 2001 To err (meiotically) is human: the genesis of human aneuploidy. Nature Reviews Genetics 2, 280-291.
6. Beatty, R. A. 2008 The genetics of the mammalian gamete. Biological Reviews 45, 73-117.
7. Wilmut, I., Sales, D. I. & Ashworth, C. J. 1986 Maternal and embryonic factors associated with prenatal loss in mammals. Reproduction 76, 851-864.
8. Bloom, S. E. 1972 Chromosome abnormalities in chicken (Gallus domesticus) embryos: types, frequencies and phenotypic effects. Chromosoma 37, 309-326.
9. Forstmeier, W. & Ellegren, H. 2010 Trisomy and triploidy are sources of embryo mortality in the zebra finch. Proc. R. Soc. Lond., Ser. B: Biol. Sci.
Monday, November 08, 2010
A botanist once told me...
Botanists have to make up dirty rhymes because they can't just take fecal samples the way zoologists do.
Monday, November 01, 2010
Halloween, diversity, and the left-leaning scientists
One could easily suggest several possible reasons for left-lean. The liberal might say that scientists are people trained to think carefully about ideas, and liberal ideas stand up to careful thought better than their conservative counterparts. She could add that conservatives, particularly the United States, tend to engage in a great deal of anti-intellectualism, which is not a good way to win the support of scientists and that conservative leaders and movements twist scientific conclusions and ideas more frequently and more disastrously than do liberals. A conservative could argue that as a big-spending liberals tend to support science funding, scientists have a direct interest in supporting liberals. I think all of these things are at least sometimes true.
Another hypothesis occurred to me last night during our Halloween party. Looking around our apartment, I saw not only several Americans (tiger, mime, fisherman, pirate, little girl/trick-or-treater, scarecrow, frog, Dorothy, lumberjack, but no tricorn hats) and Germans (lizard, clown, flapper, schoolgirl, bus stop, Little-Red-Riding-Hood, ninja, and a little boy dressed as a little boy), but also friends from Italy (a mouse and a mummy), Hungary (bank robber), Austria (ghost and witch), Australia (tin man and geisha), Poland (burglar, witches and a ghost), Latvia (witch), Spain (ghost and sorceress) and Finland (clown, penguin, bear and ladybug). We had 34 people from 10 countries, and had invited people from at least a dozen more (Japan, Taiwan, China, Mexico, Brazil, the Netherlands, Denmark, England, Russia, Bulgaria, Turkey and New Zealand). This sort of national diversity is common at gatherings of Institute researchers. I had a similar experience at Berkeley, with colleagues from all over the world.
Conservatives in many countries are nationalistic, or at least view the traditional ways of doing things in their part of the world as best and most correct. So perhaps the conservative hoping to build a career in science finds herself in many uncomfortably international situations, where her assumptions are challenged or potententally unpopular. If so, such people may tend to either revise their opinions (or pretend to, which often eventually leads to an actual change) or avoid such situations. Such avoidance would make a successful career in science difficult, at least at the prestigious institutions which tend to have international research staff.
The Ivory Tower's relative lack of political diversity may partly result from its high demographic diversity. It is hard to condemn witches when your living room is full of them.
Monday, October 18, 2010
Cold Season
it hovered in a frozen sky, and gobbled summer down."
-Joni Mitchell
Rostock has its first real frost this morning. In anticipation of this event, the first bad cold of the year has been going around. I think about half the Institute has gotten it so far. I have been sick to varying degrees for a week now. Without having actually looked at the relevant research, my understanding is that we get so many more colds when it gets colder because the airborne viruses break down much more slowly when the areas cold and dry and sunlight is weak. I've also heard somewhere that the cold air makes mucous membranes more susceptible to viruses. This all makes sense, and helps explain why that other common airborne virus that spreads every year, the flu, also concentrates in winter, but it leaves me wondering this: is the pattern the same in species that are adapted to highly seasonal climates? Humans are basically a tropical species that construct tropic-like microclimates for ourselves wherever we go. Our ancestors a few thousand generations ago would not have experienced the yearly cold season as we do today. Moose on the other hand have been living in cold climates forever. Their bodies should expect high virus conditions every winter and prepare accordingly. I speculate that the immune systems of such animals are seasonal, being better at fighting airborne viruses in the winter, and perhaps skin fungus is in the summer. I wouldn't personally want to do the experiments to find out, but I would read the paper if somebody else wrote it.
Saturday, October 16, 2010
Not buying a PC
Friday, October 15, 2010
The LPU
The Least Publishable Unit has a long and proud history in science. It is often the case that one can either toil for months over a very long paper, or chop that up into several smaller papers which will come out faster, often in lower profile journals. The quality of the work is not necessarily any lower, but the step is more incremental and the CV filled out faster. This particular LPU is a simple reanalysis of some data from my doctoral work. I started writing it today, and expects to have completed draft by sometime next week. I will submitted to a low-profile journal with rapid turnaround and hope to have an answer from them before I send in my CV.
I do not feel at all bad about this, my first LPU, for several reasons. First, looking through the CVs of most of the very successful scientists I know, there are quite a few little papers among their giant publications. Second, I can think of several papers which seem to me to have started out as LPUs but which turned out to be tremendously important and widely cited. Third, experience and mentors have told me people are more likely to read a short paper. Fourth and finally, writing short papers is a hell of a lot easier when one has to dictate everything.
There is one way in which this paper does not really meet the classical definition of an LPU: there are lots of data behind it. The true LPU should have just enough data to make a publishable paper. In this case, the sample being analyzed is fairly enormous, although not much bigger than is needed to answer the question.
All in all, writing a little paper seems a good change from the massive review article I've just submitted. I hope it turns out to be as easy as I think it will.
Friday, October 08, 2010
Big grant, small grant application
Assuming I can convince them of these things, more so than the many other applicants, it will be a pretty sweet deal. They will not only give me a significantly increased salary for five years and enough funding to get my experiments going, they will pay for me to hire a couple of graduate students and a postdoc or two. Frankly, this is more of a career advance than I think I am likely to receive at this point.
The nice part, other than the generosity of the award should I get it, is how little work they ask of me for the application. They want only a one-page statement of my Scientific Accomplishments (I'm not sure what these are yet) plus 2 pages of Research Plans. I could give them 20 pages of research plans with little difficulty if my hands worked well, but under the circumstances I'm much happier to give them 2.
How my health issues will work into this whole application is an interesting question. There is no doubt that I would have been more productive this year and in grad school had been healthy, but I doubt that they can or should take this into consideration. The best I can hope for is that one of my letter writers will mention something about dedication to science or gumption or the fact that I just keep coming back, like a bad case of poison ivy.
On a more philosophical level, I can ask this question: if someone has a disability which interferes with their productivity, and is likely to continue interfering with their productivity, should an employer consider how productive the worker would be without the disability, or should they simply asked of each candidate, "how productive is he/she likely to be?" I would like to say the former, but from the employer's point of view, it's hard to make the case against the latter.
On the other hand, my joint problems potentially make me better qualified to think of the questions and tell other people to gather the necessary data to answer them than I am for actual data gathering and analysis. The higher they promote me, the more qualified I may be.
Friday, October 01, 2010
The news from me
I had carpal tunnel surgery on my left hand about a month ago and am pleased to report that had very little numbness or tingling since then in that hand. I still have pain around where the incision was and some of the internal cuts but they assure me that will fade with time. I'm having the same surgery on my right hand on Monday. I'm supposed to be on sick leave for all of next week, but I suspect I'll end up coming in to use the dictation software, as I don't have it at home (I have a Mac it works on PC.) I hope that by the end of the year I'll be able to use my hands fairly normally again.
I also just found out today that a large grant application I need to submit is due on November 17. This may interfere somewhat with my plans to post here more regularly.
Fall
Wednesday, June 16, 2010
experimentation
Thursday, May 27, 2010
Look Ma, ...
There are many jobs for which not using one's hands would be a greater problem than they are for me as a scientist. A carpenter, a cellist, or a cashier would be much less able to work around this problem and I am. Furthermore, there is extremely little short-term pressure for me to get anything done. In the long term, I have to publish papers to keep my job and move to better jobs in the future. In the short term I want to get things done simply because it is too frustrating to not do so.
I'm using dictation software which, now that I've used for some time, is quite quick and accurate for creating text. Not quite as fast as typing, but a hell of a lot faster than I thought dictation software was before I started using it. I have students to do my lab work for me, I'm not doing any field work, and I have access digitally most of the literature I need. Given all this, why should a small matter like hands make much of a difference?
In practice, there are all sorts of things that I find myself unable to do. The dictation software supposed to make it possible to use many different programs, for surfing the web, analyzing data, sending e-mails and so on; in fact many of these thing are quite difficult to do without moving the mouse or touching the keys. Writing code is extraordinarily difficult. Reading heavy paper books is quite hard. Many software tools are simply inaccessible.
My latest stratagem is to order a mouse controlled by foot. The ability to point and click should solve many of my problems, in combination with this dictation software.
The other challenge of being productive despite my hands, is one of concentration. Doing things so differently requires a lot of thought about how I do them, which distracts me from the ideas and tasks to concentrate on. In addition to that, my hands hurt, tingle, throb and otherwise distract me. I am frankly not so good at ignoring it to the point that I can think deeply.
Ultimately, my hands will probably get better with time and coddling. When that happens, I can add dictation software and foot-controlling a mouse to my long list of random and not particularly useful skills. Until then, I'll be studying the list of available voice commands and wearing slip ons.
Sunday, April 18, 2010
Planeless sky
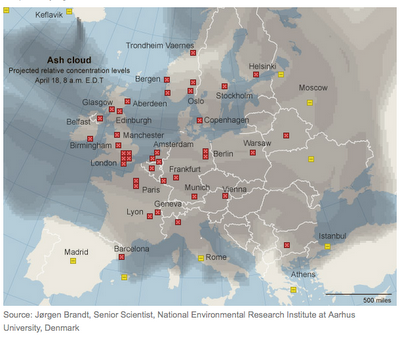
This map (from the NYTimes) shows which European airports are currently closed. Rostock is right in the middle of the gray area, half way between Berlin and Copenhagen. The closest operating airports right now are Madrid (2500km), Rome (1700 km including crossing the Alps) or St. Petersburg (2 days by ferry), and they are all booked up. So I may not have many coworkers around until this dust blows over.
The sunsets have been colorful the last couple of days, and the horizon looks hazier than usual, but other than that and the lack of airplanes flying over, I can see no smoke. Also, the grocery stores are out of bananas.
The airlines are starting to question the science behind the flight ban. I am doubtful myself. The concentration of particles (which hasn't actually been measured in most places) must be pretty minute, as the emissions from a small eruption are dispersed over perhaps 100,000,000 cubic kilometers.
I come to that rough estimate because the effected area is about 5000km east to west, 5000km north to south and the cloud is about 4 km top to bottom. I've not seen an estimate for how many tons of ash are up there, or what concentration is dangerous, but I'm guessing the concentration of particulate matter up there is no higher than that over LA or Houston in the summer.
Wednesday, April 14, 2010
Die Fledermaus
little posting
The following is from the
PLoS Biology Editorial and Publishing Policies
"PLoS Medicine, PLoS Biology and PLoS ONE do not consider for publication papers where any of the research costs or authors' salaries have been funded, in whole or in part, by a tobacco company. "
Discuss
Sunday, March 14, 2010
Evolutionary models of evolution
As an evolutionary biologist, I would like to encourage those modeling the evolution of aging to include in their models actual evolution. Optimization models and even Markov Chain models, while very useful, are not evolutionary. By this I mean that they do not include populations changing through descent with modification. To meet a biologist's definition of evolution, a process must include individuals who are reproducing and the offspring must be modified copies of the parents. This requires a population of individuals with heritable traits and mutation rates which modify the parents' traits in the offspring. In order for adaptive evolution to occur, these heritable traits must also influence how many copies of its genome each individual passes on to the next generation.
Markov-chain models, while somewhat closer to evolutionary, still lack the aspect of a population, which is essential for evolution. In many cases the outcome of evolution will depend on having competing or interacting sets of genes within the same population. This cannot be meaningfully understood if the whole population is assumed to have only one set of genes at any one time.
A truly evolutionary model of aging must therefore be fairly complex. It must simulate individuals, who have age-specific mortality and fertility probabilities. These age-specific schedules must be determined by a set of genes. These genes in turn must be determined by a process of inheritance and a process of mutation.
Using such a method, we can address questions that are difficult to get at through optimization. For example, suppose we would like to know why closely related populations have similar patterns of aging, even when they live in different habitats, or occupy different niches. This pattern has been observed in comparative data and comes under the heading of phylogenetic inertia. With an evolutionary simulation, we can impose environments which mediate the relationship between the genes and the demography. We can then ask what characteristics of the environment or what characteristics of the relationship between environment and demography would allow the starting point (that is the initial genes and demography of the population), to influence the ending point (that is the genes and demography the population ends up with).
To take another example, optimization models generally lack any information on the structure of the genome or the process by which that genome changes. However, genomic structure and mutation process are not irrelevant to what demography the population evolves. An evolutionary simulation will allow for modification of the genomic structure or the mutations process. Compare for example, two populations, each of which has a genetically controlled pattern of investment in various tasks such as reproduction, repair, growth or immune function. In population A, as many genes control this at the beginning of life as at the end. In population B, many genes control the pattern of investment early in life, while relatively few are still influencing late life investment. In both populations the genes affecting these investments are subject to mutational pressure and to selection. In each a mutation-selection balance will emerge, but these mutation selection balances will differ between the two populations. The two populations living in the same environment will arrive at different demographies, each nonoptimal.
These are but two of the many complicating factors which can be explored using an evolutionary simulation and are difficult to get at in a model that does not include explicit evolution. Of course models should be simple enough that one can figure out what factor is influencing what outcome. A model cannot include every complicating factor biologists might like to throw in. As such, I propose a modular evolutionary simulation. By this I mean we start with as simple a model as we can which still has real evolution going on and we write it in such a way that one can add more complicated processes. For example, the basic model could have an extremely simple process of mutation, but could be written such that that this process is easy to remove and replace with a more complicated mutational process. Reproduction could be clonal, but again that process of inheritance could be coded such that it could be pulled out and replaced with sexual reproduction by someone who is interested in what effect the mode of reproduction would have on the evolved demography. The environment could be extremely simple and replaceable with a more complicated environment. I am a slow and inexpert programmer but I imagine that it would not be impossible to write such a simulation in a way that genome, inheritance, mutation, environments, and demography are interacting pieces which can be replaced as one replaces the batteries, bulb, wire, switch and casing of a flashlight. One need not modify the casing to replace acid batteries with rechargeables, or replace rechargeables batteries with lithium rechargeables. One can swap a white bulb for a yellow one without modifying the wires or switch. A properly designed base simulation would allow each of us to experiment and still be able to compare our results without any one model becoming unnecessarily complex.
Saturday, March 06, 2010
Kudzu Power
We determined the amount of standing biomass of kudzu (Pueraria montana var lobata) in naturally infested fields in Maryland and Alabama, USA. At each site, we evaluated the carbohydrate content of roots, stems, and leaves. For a third site from Georgia, we evaluated the carbohydrate content of kudzu roots of varying diameters. Belowground biomass in Alabama exceeded 13 t ha−1, and contained an average of 37% fermentable carbohydrates (sucrose, glucose, and starch). Roots from Georgia of all size classes contained over 60% fermentable carbohydrates. Biomass and carbohydrate levels in roots from Maryland were low compared to plants growing in Alabama and Georgia, producing 5 t ha−1 of roots with 20% non-structural carbohydrate. Stems from Alabama and Maryland contained 1–3% carbohydrates. Based on the yield and carbohydrate content, we estimate wild kudzu stands in Alabama and Georgia could produce 5–10 t ha−1 of carbohydrate, which would rival carbohydrate production from maize and sugar cane fields. If economical harvesting and processing techniques could be developed, the kudzu infesting North America has the potential to supplement existing bioethanol feedstocks, which could be of significance to the rural economy of the southeastern USA.
Further consider this economical processing technique, from next month's PNAS:
Abundant plant biomass has the potential to become a sustainable source of fuels and chemicals. Realizing this potential requires the economical conversion of recalcitrant lignocellulose into useful intermediates, such as sugars. We report a high-yielding chemical process for the hydrolysis of biomass into monosaccharides. Adding water gradually to a chloride ionic liquid-containing catalytic acid leads to a nearly 90% yield of glucose from cellulose and 70–80% yield of sugars from untreated corn stover. Ion-exclusion chromatography allows recovery of the ionic liquid and delivers sugar feedstocks that support the vigorous growth of ethanologenic microbes. This simple chemical process, which requires neither an edible plant nor a cellulase, could enable crude biomass to be the sole source of carbon for a scalable biorefinery.
Friday, March 05, 2010
neologistic challenge
So I want a word that can be defined as "the decline in mortality risk with age from conception to maturity." A root that means improvement, growth, blossoming, development, growing up, or something along those lines would be best. Twelve points for whoever comes up with the best term.
Saturday, February 06, 2010
Cold and Fat
This simple idea has several profound implications that I will now proceed to invent.
The first implication is that " burn lots of energy to stay warm" means something very similar to "increase metabolic rate." So people in colder environments should have higher metabolisms. This matches with the current experience of myself and my wife. For the past months the temperature outside our apartment has rarely gotten above freezing, the wind is always blowing and it is generally damp. Our apartment is somewhat difficult to heat, so it is generally cool and sometimes downright cold inside. We have been eating a very rich diet, and rather than gaining weight, I think we are both loosing a bit. And although the short days cause a degree of lethargy, I have been generally quite productive, with fewer problems of concentration than usual.
There is good reason to think, in fact, that colder climates lead to greater productivity. Colder countries are systematically more economically productive than hotter countries, air conditioning raises productivity considerably, and hot countries experience more economic growth in cool years than in warm ones. My preferred speculation is that this is because the experience of coolth induces greater physiological throughput, allowing for greater activity. If one needs to expend energy to keep oneself warm, why not put that energy to some good use, such as thinking, building, or working. Why shiver when I can use the same energy sharpening the knives or generating a hypothesis?
Allow me one further observation and conjecture. Germans, who eat very heavy diets but on the average are less heavy than Americans, are in the habit of opening all the windows whenever it gets warm inside, even if it is below freezing out. Two apparently unrelated stereo-types of modern Americans, both of which have some basis in fact, may in fact be causally related. These are, we are very fat, and we keep our houses very warm in the winter. Perhaps the miracle diet so many have been searching for should include turning down the thermostat. If we burn more calories when we experience cold, and we want to burn more calories, perhaps we should experience more cold.
Wednesday, February 03, 2010
A month of snow
Friday, January 29, 2010
Sprouting collaborations
Here's the recipe:
Wash the sprouts, peel off any out leaves that don't look good, and cut each sprout in half.
Cut up a head of garlic.
Juice 2 lemons.
Heat a large cast-iron skillet or other frying surface, large enough to fit all the halved sprouts in a single layer. Put on a fair bit of oil and bring to very hot. Pour in the sprouts and use a spatula or fork to flip them over so that the flat side of each is down. Slide them around some so they all cook evenly. Cook them like that until they are somewhat charred on the bottom. Then flip them over and roll them around so that the other surfaces get cooked. They will still be somewhat too hard inside, so pour in a scant cup of water and cover while the water rapidly boils off, steaming the insides. Once the water cooks off, taste a couple of sprouts and if they are still too hard inside, pour on a little bit more water. Once they are done, remove them to a large bowl and put the garlic on to fry. After a minute, pour some splashes of soy sauce over the garlic, and let it cook down a little bit. Mix this with the lemon juice, salt and pepper, and pour it over the sprouts. Serve hot.
Wednesday, January 27, 2010
The flapper in winter
My colleague, who also watches birds came into my office to ask if I had noticed it, and if so what I thought it was. He said there were footsteps in the fresh snow near it. He was right. Through my binoculars I could see swan prints leading to where it sat. And that lump on top might be a bit of neck leading to a head tucked entirely under a wing. But it must be dead I thought, why else would it sit so still for so long in such a windy spot on the ice?
I glanced at it occasionally though the morning, and it didn't move. Then some people walking their dog passed by, and the dog made like it was going to run out onto the ice to get the swan. A long gray neck snaked out from under the wing and looked straight back at the dog. The dog must have realized the ice was too thin to hold it. As soon as the dog was gone, the head disappeared under the wing again, and there it stayed.
In the early afternoon a fire department rescue truck pulled down the road to the harbor near the swan. Two guys, one holding binoculars, got out and looked at the swan for a while. I wondered if they thought it was a child. They drove away and came back with a third guy, who threw snowballs near the swan and yelled at it until it got up and walked a few steps. They left again, and the swan sat back in its usual spot, head under wing. The snow got heavier and started to pile up on the windward side of the swan, but it stuck to its spot, and still was there when the sun went down.
Monday, January 25, 2010
Almost finished
Friday, January 22, 2010
I really didn't want to get into discussing testis size.
Monday, January 11, 2010
Lovers in the snow
But in the tree in front of my office window, two magpies are carrying fresh twigs into a crook between three branches. There is only one reason I know of why they would be doing this: they are building a nest. Most birds, including magpies, only build nests to lay eggs in, and now is not the time to lay eggs. It is too cold for the eggs to develop (even with mom sitting on them), and if they did hatch , there would be nothing to feed them. Any nest built now isn’t even likely to still be in good enough shape to use come spring. The spot where they are putting the twigs is near the top of the tree, on the branch closest to the river, and shakes whenever the wind blows, which it does frequently.
This raises the question of why? When other birds in the neighborhood are struggling just to keep from freezing or starving, why are the magpies wasting their time and exposing themselves to the cold building a nest they can’t use? Perhaps the cold has driven them mad? Maybe they are pulling food out of the dumpster of the near by grocery store, and having plenty of food, think it is time to breed? A genetic disorder?
There may be some perfectly good reason for this (pair-bonding activity?) but I’m not sure I buy that.
When my colleagues and I were writing a paper on the definition of behavior, many of previously published definitions we came across specified that behavior is adaptive, that it will tend to increase the fitness of the individual performing the behavior. We omitted this from our definition, because there are so many behaviors whose adaptive significance is uncertain, or which seem maladaptive. It is certainly true that most behaviors are adaptive, some, like building a nest in a snow storm, probably are not.
Wednesday, January 06, 2010
Rostock ten day forecast
Snow
Mostly Cloudy
Snow
Snow
Snow
Snow
Snow
Snow
Snow
I must really love science, as I don't know what else would keep me in this sun-forsaken place.
Sunday, January 03, 2010
Picking sources
I and my assistants at Berkeley classified a significant number of primate species based on how much care fathers give to their offspring. For each species we would search the primary literature for sources documenting if fathers in that species provided care to their offspring, and if so how much. We gathered papers, sometimes dozen for a single species, read the relevant pieces and decided that fathers of that species provided no care, little or incidental care, significant care or were, for at least some part of their offspring's lives, the primary caregivers. This compilation allows me to compare the evolution of paternal care with that of other traits, such as the degree to which males fight for access to females in each species (the prediction being that males will tend to either invest heavily in their offspring or fight intensively for mates, but not both).
But what I have to do now is decide, for each species individually, which paper or papers to say I based my decision on when I publish this compilation. I've not actually studied any live primates myself. The many people who have dedicated years of their lives to documenting the behavior of each species, may disagree with my conclusion on their species (some inevitably will, I hope not too many). Those that do will be prone to dismiss the paper as a whole unless I at least give appropriate references to show I wasn't just making my data up. I feel like somehow I've made it this far without really knowing what rules to follow in documenting my sources. One can't just list every paper one consulted, or pick one at random. Should I reference the first paper to suggest that this species had that trait? Or the one with the strongest evidence, or the review article that states that the evidence for that conclusion is overwhelming? Should I include a reference to the dissenting papers, to show that I am aware of them, even if I disagree? Should I try to include references from the journal I plan to submit to?
What I'm actually doing, which may be the wrong approach, is referencing the papers I find most convincing and relevant, regardless of precedence, author or journal. As long as the reviewers don't object, I'll consider this strategy a success.
Saturday, January 02, 2010
To survive and reproduce in good times and bad
Is there reason to believe, or evidence to support, that the forms of evolution occurring among species during a period of abundant resources is different from the forms of evolution occurring among species during a period of deficient resources?
It seems that a lot of the argument in evolutionary theory is that it takes a lot of energy to grow extra and useless appendages or what have you, so if they really are useless, you'd expect them to evolve away. But if resources are abundant--energy is not a particularly limiting factor--do you then get a scenario in which all kinds of wacky and useless appendages appear and are not attritioned away? Which gives those appendages time to hang around enough to be available when the environment changes and all of a sudden they are useful and confer an advantage?
Or something like that?
I wouldn't go so far as to say the "forms of evolution" are different. In good times and bad evolution acts through natural selection, genetic drift, mutation and all the same basic mechanisms. Rather I would say that selection acts of different traits, or favors different forms of those traits, depending on if times are good or bad. One excellent example of this has been documented by Peter and Rosemary Grant in long term studies of Darwin's Finches on the Galapagos Islands. The climate in the Galapagos is impacted hugely by the El Nino/La Nina climate cycles. In some parts of the cycle, the islands are cool and damp, vegetation grows lush, and there are lots of big seeds to be had. In other parts of the cycle, it is very hot and very dry and only the desert plants with their tiny little seeds are producing. In the good years, the finches with the big bills can eat lots of big seeds, and reproduce like mad. In only a few years the population of one finch species is dominated by big-billed finches. Then when the rains stop, the population starts to crash, and the finches with the little bills good for extracting and opening small seeds are much more likely to survive. After a few years of that, the population of that same species is again dominated by small billed finches. This isn't individuals developing differently depending on the food supply, this is just massive, cyclical natural selection driving the population's genetic make-up around in circles.
On a much larger time scale, generalists are much more likely to survive large extinction events, while specialists often dominate in habitats that have been very stable for millions of years. Consider which of each of these pairs of species is in greater danger of extinction?
German Cockroaches or Lord Howe Island Woodeating Cockroaches
The Black Rat or the Salt-Marsh Harvest Mouse
The Common Pigeon or the Mariana Fruit Dove
The Common Raccoon or the Cozumel Raccoon
Goats or Alpine Ibex
Humans or Sumatran Orangutans
In each case the generalist are doing fine, while their specialized relatives can't cope with change. The fossil record shows multiple examples of large groups going extinct when the coprolites hit the fan, but one or two very generalized species in those groups making it through and giving rise to many new species. The amazing thing is that over and over most of those new species are specialists, evolving to be increasingly good at dominating increasingly narrow sets of resources. Give Rattus rattus a few tens of millions of years and no other mammals on the planet, and they would evolve into many thousands of separate species, filling a vast array of niches, and most of those species would be specialists. If another great collapse came, the ones most likely to make it through would again be the super generalized rat.
As far as the "useless appendages" argument goes, remember that even when resources are abundant, there is still the race to see who can convert those resources into the most offspring the fastest. Plus, the ideal situation rarely lasts very long. Usually within a few generations the population of predators has increased, the food supply has diminished, or population density has gotten so high that pathogens are spread easily. Exponential growth is not to be underestimated. So with the possible exception of humans over the last couple of hundred years, it is almost never the case that a population goes on growing for many generations without selection knocking back those who spend their energy recklessly.
That said, there are traits that are advantageous in bad time and costly in good times, or the opposite. Sometimes species evolve plasticity, such as the ability to grow a thicker coat when the winter is colder, but not waste the protein in mild winters. And sometimes, like Darwin's finches, they just evolve back and forth. The camel's hump is probably something of a hindrance when water and food are plentiful, but it bears that cost because more often than not things will get dry again, and that hump will save its life. If camels lived in an environment where it didn't get dry for some thousands of years, they might end up sans hump, looking more like big llamas. Or they might just die out, vanquished by cows and goats. Camels, after all, are specialists.
Thrice bitten
1. I have been stung by a Portuguese Man o'War.
2. I have been poisoned by a black widow spider.
3. I have been bitten by a venomous snake.
The third was the one I intended to be false. I have just found out that all three of these are true. The only snakes I have ever been bitten by are those I have picked up, and the only snakes I have picked up are those which I knew to be non-venomous, primarily garter snakes. It turns out though, that garter snakes are venomous, and my knowledge (shared with most of the rest of the world) of their lack of venom was wrong. Mind you, they almost never actually get any venom into a human, and if they do it is not dangerous. But it turns out garter snakes do have venom glands, and of the dozens I've handled I've twice been bitten by them. Once picking up a tiny individual from under a rock, and once rescuing a larger individual from a road. They don't have fangs, or dental groves, or other efficient venom delivery systems, and their venom isn't strong enough to kill even small prey, but apparently if they chew on a prey item long enough some of the poison will work its way into the wound and perhaps help to subdue the food, easing swallowing. I once watched a small garter snake attempt (for about twenty minutes) to swallow a very large frog, and I must say the poison didn't do the snake much good, despite extensive chewing, because the frog escaped after giving the snake a good kicking about the head.
If I'm ever forced into that game again I will have to change my lie to, "I've been bitten by a venomous mammal." I'm not dumb enough to pick up a shrew.



